Singclean COVID-19 Igg/IgM Test Kit (Colloidal Gold Method)
- Location: China
- FOB Price: ( Negotiable )
- Packaging Detail: As per standard
- Delivery Time: 35 Days Day
- Payment Type: L/C, T/T
Product Description
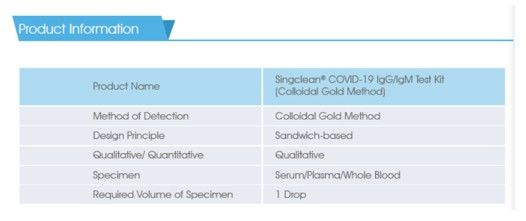
CE Approved Covid-19 IgG/IgM Antibody Blood Test Kit can be used as an initial screening for qualitative and differential detection. Throughout testing the antibody of IgG and IgM immunoglobulins in human serum, plasma or blood, the test kit can show the result in a short time.
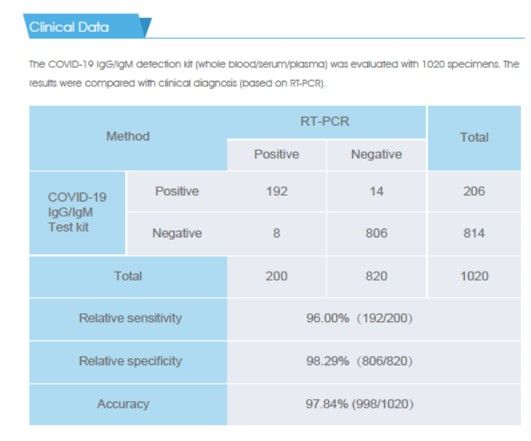
Relative sensitivity 96.00%(192/200)
Relative specificity 98.29%(806/820)
Accuracy 97.84% (998/1020)
Advantages
Fast detection: 10 minutes to interpret the result;
Easy to operate: no complex equipment required during test;
Sensitive: the product has a higher sensitivity than PCR Nucleic Acid
Global Network
Singclean@COVID-19 lgG/gM Test Kit has been sold to more than 50 countries in the worid, like Brazil, Chie, Poland,spain, Bolivia, Greece, Lihuania, Ecuador, Ukraine and Turkey.
Precaution
It is worth noting that the result of Covid-19 IgG/IgM antibody blood test should not be used as the only evidence of Coronavirus diagnosis. Professional doctors and clinical findings are necessary if the result of antibody test is positive. Besides, the test kit should be operated by professionals only. Use by individuals may cause deviation in test results.
Limitation
False negative and false positive could happen. If the antibody is lower than the detection level, especially in the 7 days of the onset of symptoms, a positive patient may also be diagnosed as negative.


Relative sensitivity 96.00%(192/200)
Relative specificity 98.29%(806/820)
Accuracy 97.84% (998/1020)
Advantages
Fast detection: 10 minutes to interpret the result;
Easy to operate: no complex equipment required during test;
Sensitive: the product has a higher sensitivity than PCR Nucleic Acid
Global Network
Singclean@COVID-19 lgG/gM Test Kit has been sold to more than 50 countries in the worid, like Brazil, Chie, Poland,spain, Bolivia, Greece, Lihuania, Ecuador, Ukraine and Turkey.
Precaution
It is worth noting that the result of Covid-19 IgG/IgM antibody blood test should not be used as the only evidence of Coronavirus diagnosis. Professional doctors and clinical findings are necessary if the result of antibody test is positive. Besides, the test kit should be operated by professionals only. Use by individuals may cause deviation in test results.
Limitation
False negative and false positive could happen. If the antibody is lower than the detection level, especially in the 7 days of the onset of symptoms, a positive patient may also be diagnosed as negative.

Contact Information
- Company: Hangzhou Singclean Medical Products Co., Ltd.
- Address: Hangzhou, Zhejiang, China
- Telephone: 86-18806512950
- Mobile: 86-18806512950
Company Profile
- Hangzhou Singclean Medical Products Co., Ltd.
- [ China ]
-

-
Hangzhou Singclean Medical Products Co., Ltd. was established in 2002, and currently has 18 years of expertise in the industry of absorbable biomaterial with nearly 400 employees. The production base covers an area of 28,000 square meters, with 8 GMP purification workshops of 2,000 square meters, a logistics center of 4,000 square meters, and a R&D center and a quality inspection center of 1,00...
Basic Information
- Business Type: Manufacturing
- Company Products / Services: hyalurnic acid,covid-19 test kit,absorbable hemostatic gauze,absorbable hemostatic particles,dermal filler,hyaluronic acid skin care products
- Year Established: 2002
- Number of Employees: 101-500
- Website: http://www.singclean.net/